#SootinClaimon.Com : ขอบคุณแหล่งข้อมูล : หนังสือพิมพ์ The Nation.
First coronavirus vaccine shipments set to arrive in states Monday (nationthailand.com)
First coronavirus vaccine shipments set to arrive in states Monday
Health & BeautyDec 13. 2020 Pharmacy Director Ahmed El Kority opens a new ultra freezer that will be used to store COVID-19 vaccines at the Riverside Shore Memorial Hospital in Onancock, Va. MUST CREDIT: photo for The Washington Post by Parker Michels-Boyce.
Pharmacy Director Ahmed El Kority opens a new ultra freezer that will be used to store COVID-19 vaccines at the Riverside Shore Memorial Hospital in Onancock, Va. MUST CREDIT: photo for The Washington Post by Parker Michels-Boyce.
By The Washington Post · Frances Stead Sellers, Ariana Eunjung Cha, , Lena H. Sun, Isaac Stanley-Becker · NATIONAL, HEALTH, HEALTH-NEWS
WASHINGTON – Hospitals that have spent months seeking a silver bullet against a virus that has killed more than 295,000 people in the United States will begin receiving shipments of the first coronavirus vaccine on Monday, U.S. officials said, comparing the start of distribution this weekend to the Allied invasion of Normandy in June 1944.
Saturday, said the four-star Army general overseeing vaccine rollout, was “D-Day,” following the Food and Drug Administration’s Friday-night clearance for emergency use of the two-dose regimen developed by Pfizer and the German company BioNTech.
“D-Day was a pivotal turning point in World War II; it was the beginning of the end,” said the general, Gustave Perna, who is chief operating officer of Operation Warp Speed, the public-private partnership speeding the development of vaccines and therapeutics. “D-Day was the beginning of the end, and that’s where we are today.”
The initial distribution of 2.9 million doses, a sliver of what was initially anticipated and intended only for health care workers and residents and staff of long-term care facilities, will arrive at hospitals battling climbing case counts and mounting deaths. Immunization in its early phases will not curtail intensifying outbreaks, experts cautioned, underscoring the need for continued public-health precautions.
But the vaccine’s clearance on Friday night from the FDA, followed by backing on Saturday from a Centers for Disease Control and Prevention advisory group, set into motion one of one of the most complicated logistical missions in U.S. history, marking a new phase of the pandemic. The vaccine, which must be stored at ultracold temperatures, is being sent nationwide by plane and guarded truck.
“It’s a hugely important step,” Nancy Messonnier, director of the CDC’s National Center for Immunization and Respiratory Diseases, said Saturday as the CDC’s Advisory Committee on Immunization Practices recommended the nation’s first coronavirus vaccine for people 16 and older.
Members of the committee stressed that the vaccine, while developed in record time, had moved transparently through all required regulatory channels. They also expressed alarm about the lack of resources available to state and local authorities to carry out vaccination, in contrast to the billions in taxpayer dollars devoted to quickening the vaccine’s rollout.
As experts gave final sign-off, boxes were being packed and loaded with the vaccine, Perna said. The cargo would begin moving Sunday morning from Pfizer’s manufacturing facility in Kalamazoo, Mich., to FedEx and UPS hubs nationwide, he said.
Vaccine, he predicted, would arrive at 145 sites, mostly large hospital systems, on Monday, with another 425 sites receiving supply on Tuesday. The final 66 of the 636 locations poised to receive doses in the first round of Pfizer shipments would receive their supply on Wednesday, Perna said.
The general made clear that earlier-than-anticipated clearance from federal regulators, which took place late Friday instead of Saturday after pressure from the White House, did not alter the timetable for the distribution or actual administration of the shots. Delivery, he said, must occur when “professionals are available to receive it, and then eventually administer it,” making Monday the anticipated target.
Top FDA officials took steps on Saturday to assure the public that the vaccine was safe, that its clearance was not driven by politics and that health care sites would be equipped in the event of a rare allergic reaction observed among two British health care workers with a history of severe reactions.
Stephen Hahn, the FDA commissioner, told reporters he would get the vaccine as soon as he was eligible for it, denying, as he did on Friday, that his job had been threatened over the timing of the vaccine’s approval. Asked about the possibility of allergic reactions, Peter Marks, director of the agency’s division that regulates vaccines, said officials had examined possible side effects and concluded that people should be vaccinated unless they have had a severe reaction to one of the vaccine’s components.
The initial shipments include only the first shot in a two-dose regimen for some of the nation’s most at-risk people, with another 2.9 million doses set for distribution 21 days later. According to CDC recommendations, the nation’s 21 million health care workers and three million residents of long-term care facilities should receive the vaccine first. With a second vaccine from Moderna expected to gain approval from the FDA soon, as many as 40 million doses could be delivered by the end of the year – enough to vaccinate the CDC’s first priority group. Additional vaccines are in late-stage trials.
Health care workers, who have been prioritized because of their exposure to the virus and critical role in sustaining the nation’s strained health care system, will begin receiving the shots within days. Each hospital system is moving on a slightly different timetable, depending on resources and staffing needs, with many saying vaccination would not begin until Wednesday. Some medical centers were independently reviewing the vaccine data to double-check the FDA’s decision.
Inoculation at long-term care facilities could begin by the end of the week, Perna said. Separate kits with needles and alcohol wipes are being shipped so as to sync up with vaccine batches at each site, underscoring the complex choreography of the operation.
Perna tempered his confidence with an acknowledgment of the challenges involved in a mass vaccination campaign against a rampaging virus, with limited supplies initially available for a country of 330 million people eager to return to normal.
“We know that the road ahead of us will be tough,” Perna said.
The vaccines will arrive at hospitals overwhelmed with covid patients. As of Saturday, more than 108,000 were hospitalized nationwide, according to Washington Post data.
These facilities have spent months grappling with the ethical and logistical challenges of distributing the vaccine. At some institutions, intensive care unit staff members will receive priority; others are including those who work in housekeeping or with cancer patients or newborns. Hospitals wrestled with whether to include those working with covid-19 patients in full protective gear ahead of masked staff members interacting with people who may be asymptomatic carriers.
At many health care institutions, surveys are quietly being sent out, lotteries launched and invitations issued to one of the most highly anticipated events of 2020: the opportunity to get in line for a shot. At the same time, institutions are seeking to address unease about a brand-new vaccine.
On Thursday, Temple Health in Philadelphia emailed 3,000 invitations to employees deemed at “high risk” of infection. Within minutes, janitorial staff and anesthesiologists began signing up. And within 24 hours, each of the 252 slots at the main campus’s vaccination site was taken.
High demand assuaged Chief Medical Officer Tony S. Reed’s fears that vaccine hesitancy would keep people away, wasting precious vaccine. Among the first takers, who will likely start receiving shots Wednesday, will be people working on covid-19 floors and in the emergency room. The goal is simple, Reed said: “To do the most good for the most people.”
In Philadelphia, all of the city’s hospitals will be receiving vaccine, either directly from the federal distributor or from the city health department’s ultracold storage, said James Garrow, a spokesman for the health department.
On Thursday, a package needed to administer the Pfizer vaccine suddenly showed up at the University of Pittsburgh Medical Center’s Presbyterian hospital, including vaccination record cards, masks, visors, information sheets, syringes and the diluent that needs to be mixed with every dose before it is injected.
The multi-facility Cleveland Clinic has set up a “refrigeration farm” with rows of gleaming white freezers capable of keeping the vaccine at Antarctic temperatures. The atrium at the UF Health Jacksonville hospital in Florida is being turned into a vaccination site. And other hospitals are preparing to receive the ancillary supplies needed for the finicky Pfizer vaccine.
But much depends on the supply of vaccine.
Dora Anne Mills, who is overseeing the vaccine rollout for MaineHealth, which operates 10 hospitals, said the entire system may get around 970 doses in this first round – covering a fraction of the 17,000 patient-facing employees the system aims to vaccinate as soon as possible.
“We are all facing the same dilemma: How do we stabilize the hospital systems at a time when we have so many doctors and nurses out because of covid exposure?” she said. The hospital has been holding Zoom sessions to explain the vaccine to staff and convince them of its safety.
At Salt Lake City-based Intermountain Healthcare, a 23-hospital system that serves Utah, Idaho and Nevada, officials have asked staff to sign up around their shifts in case they have mild to moderate side effects in the days after they get the vaccine.
In Minnesota, which has reported one of the nation’s highest per capita rates of infection, state health officials have meticulously planned for months the rollout of the vaccine. The Minnesota Department of Health designated 25 main distribution hubs around the state that will deliver the vaccine to 118 smaller facilities, including in rural areas.
Still, Gov. Tim Walz acknowledged lingering concerns, including access to dry ice, which the Democratic governor said could pit the state and health-care industry against Upper Midwest cheese and dairy producers that use dry ice to ship cheese curds.
In Florida, Gov. Ron DeSantis (R) said this week he expects the state to receive 180,000 doses of the vaccine in its first release. Of those, 100,000 doses will go to five hospitals that can store the shots, DeSantis said. The rest of the doses, he said, will go to nursing homes and long-term care facilities.
John Couris, president and chief executive of Tampa General Hospital, acknowledged challenges in convincing staff to be immunized. He noted, “If we say you must take the vaccine, if you don’t, what are we going to do? Terminate them?”
At the UF Health Jacksonville hospital, the atrium offers space to spend 15 minutes monitoring those who are vaccinated. The space is large enough to accommodate 500 to 1,000 people a day, said Leon Haley Jr., chief executive of UF Health Jacksonville and Dean of the University of Florida’s College of Medicine. Haley said he will be videotaped getting one of the first doses to boost confidence for others.
Riverside Health System, based in Newport News, Va., will receive 2,925 doses for five hospitals. It expects to start vaccinating personnel at the highest risk for covid-19 exposure Wednesday, including doctors, nurses and housekeeping staff, said Cindy Williams, Riverside’s chief pharmacy officer.
“There are so many moving pieces,” Williams said. The system is used to administering flu shots, but must change its clinic layout for the coronavirus vaccine to allow for social distancing and recipients to be monitored for 30 minutes post-jab.
Riverside Health System, based in Newport News, Va., will recpehilaive 2,925 doses for five hospitals. It expects to start vaccinating personnel at the highest risk for covid-19 exposure Wednesday, including doctors, nurses and housekeeping staff, said Cindy Williams, Riverside’s chief pharmacy officer.storage to an ultracold freezer or three minutes for transfer between low temperatures and thawing. The vaccine must remain in frozen storage for at least two hours if it is put back in an ultracold freezer after room temperature exposure.
The health system had previously surveyed employees before news about the efficacy of the Pfizer-BioNTech and Moderna vaccines. It found about a third of respondents were willing to get a shot. A more recent survey this week found that about 60% were interested in getting the vaccine, Williams said.
Lauren Sauer, director of operations at the Johns Hopkins Office of Critical Event Preparedness and Response, which has hospitals in the District of Columbia, Florida and Maryland, said she is concerned about the emotional burdens being placed on already exhausted health-care workers as they are turned into role models.
“We are asking them to take a brand-new vaccine and then in addition to be advocates for patients, friends, family members,” Sauer said.
“That is a lot to ask during a pandemic.”



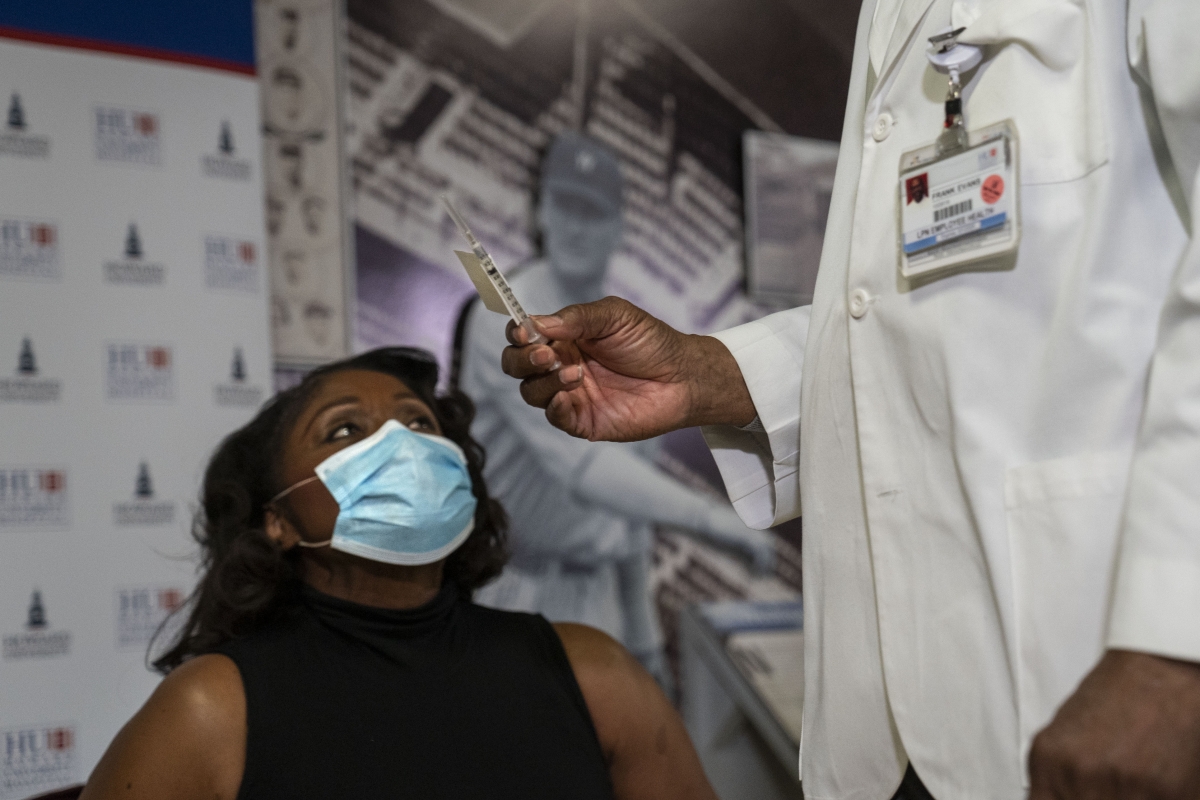
 Roy Dunlap receives the coronavirus vaccine at Howard University Hospital in Washington on Tuesday, Dec. 15, 2020. MUST CREDIT: Washington Post photo by Michael Robinson Chavez
Roy Dunlap receives the coronavirus vaccine at Howard University Hospital in Washington on Tuesday, Dec. 15, 2020. MUST CREDIT: Washington Post photo by Michael Robinson Chavez 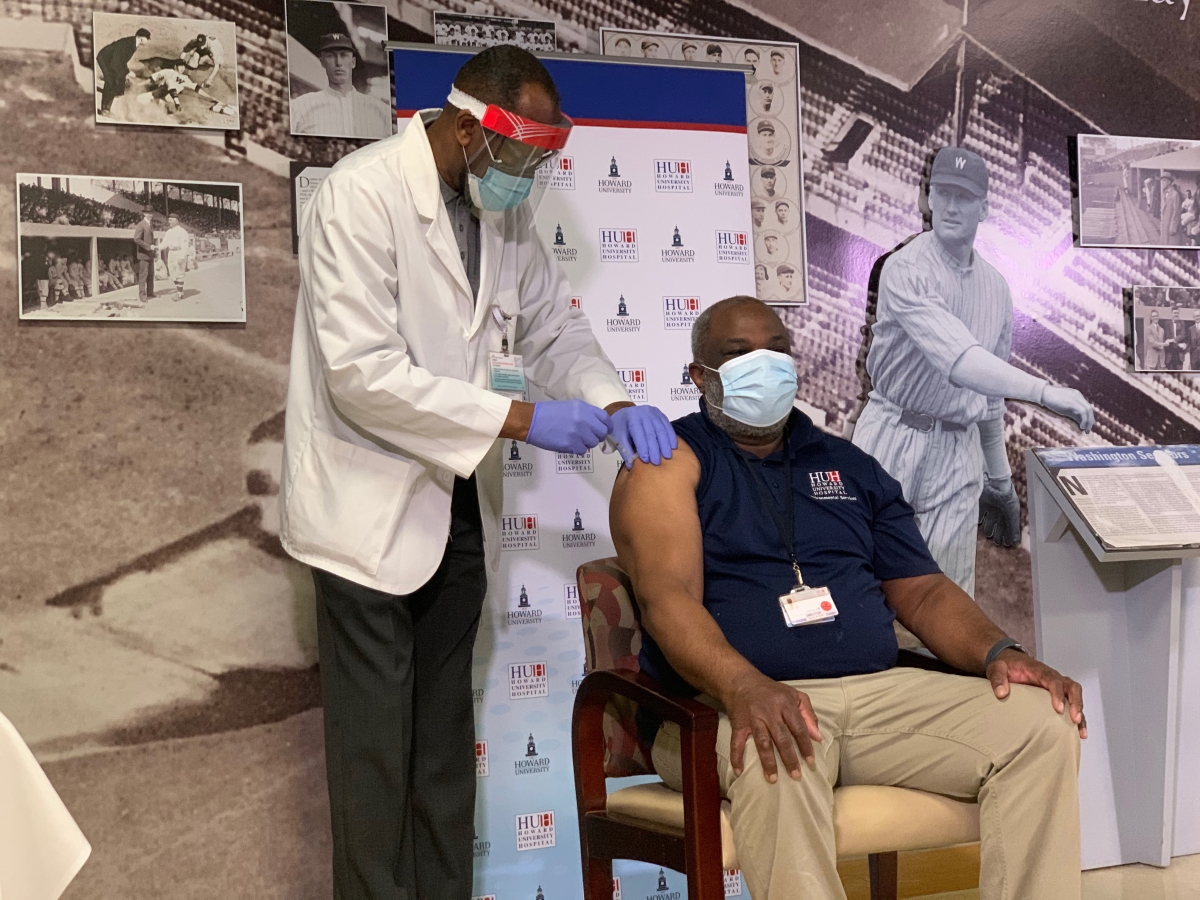
 Anutin Charnvirakul
Anutin Charnvirakul 

 Pharmacy Director Ahmed El Kority opens a new ultra freezer that will be used to store COVID-19 vaccines at the Riverside Shore Memorial Hospital in Onancock, Va. MUST CREDIT: photo for The Washington Post by Parker Michels-Boyce.
Pharmacy Director Ahmed El Kority opens a new ultra freezer that will be used to store COVID-19 vaccines at the Riverside Shore Memorial Hospital in Onancock, Va. MUST CREDIT: photo for The Washington Post by Parker Michels-Boyce.